Insights / Blog / eProtocol Automation
Clinical Trial Protocol Optimization with Gen AI: Protocol Generation for Better Results
- Abriti Rai
- September 30, 2024

On this Page
- Understanding a Clinical Trial Protocol
- What is an eProtocol?
- How AI is Advancing eProtocol Design
- Training on Real-World Protocols
- Standardized Templates and Frameworks
- Automated Document Generation
- Collaboration and Version Control
- Customization
- The Impact of AI on Protocol Generation
- Clinion’s AI: Transforming Protocol Design
- External References
- Understanding a Clinical Trial Protocol
- What is an eProtocol?
- How AI is Advancing eProtocol Design
- Training on Real-World Protocols
- Standardized Templates and Frameworks
- Automated Document Generation
- Collaboration and Version Control
- Customization
- The Impact of AI on Protocol Generation
- Clinion’s AI: Transforming Protocol Design
- External References
Protocol generation is the process of creating clinical trial protocols; the detailed plans that guide how a study will be conducted. Gen AI assists by converting a study synopsis into a structured protocol draft using standardized templates. This reduces the time and manual effort typically required, helping teams move more quickly from initial concept to a workable protocol document.
With clinical trials becoming increasingly complex, nearly two-thirds fall short of their primary objectives due to issues like poor design, flawed statistical approaches, unrealistic operational demands, limited stakeholder engagement, and regulatory challenges.
The urgency for efficient and precise protocol development has never been greater. Generative AI is a game-changer in this critical phase. By automating and optimizing protocol creation, AI accelerates timelines, enhances accuracy, and significantly lowers costs, paving the way for more successful trials.
Understanding a Clinical Trial Protocol
A clinical trial protocol is a comprehensive blueprint, describing each detail of the study. It is a critical document that defines how a clinical trial will be executed. It includes key components:
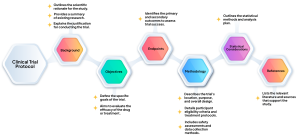
It serves as a roadmap for the trial, ensuring that it adheres to regulatory standards, safeguards participant safety, and provides a clear framework for all involved parties, including researchers, sponsors, and regulatory authorities.
What is an eProtocol?
An eProtocol is an electronically generated version of the clinical trial protocol. Digitization of the protocol creation allows for better collaboration, version control, and efficiency.
By incorporating Large Language Models (LLMs) and Gen AI, trial data from relevant sources can be extracted and used in accurately filling the protocol document’s necessary sections, thus reducing the manual effort, time, and resources spent.
How AI is Advancing eProtocol Design
Developing research protocols typically takes 160–220 hours due to its complexity and stakeholder collaboration. AI tools like eProtocol reduce this to just 1 day for generating content, with 1–2 weeks for review and styling, streamlining the process and minimizing delays.
Training on Real-World Protocols
AI-powered tools are reshaping protocol development by leveraging data-driven insights and continuous learning for greater accuracy and efficiency.
Data Collection: AI tools are trained on extensive datasets of clinical trial protocols, including study objectives, endpoints, methodologies, and regulatory requirements.
By learning from these datasets, AI generates protocol components that meet industry standards and address common challenges. GenAI, requiring less data than traditional ML models, can be fine-tuned with just a few past protocols to deliver precise, industry-standard results.
Continuous Learning: AI systems can continuously improve by learning from new data and user feedback. This iterative learning process helps the AI adapt to evolving industry standards and practices.
Standardized Templates and Frameworks
AI tools often utilize standardized templates to ensure consistency and compliance across protocols. Industry organizations develop these templates to provide a framework that aligns with regulatory requirements and best practices.
Template Utilization: The AI tool incorporates standardized templates from industry leaders like TransCelerate to efficiently structure protocols. Utilizing these pre-defined frameworks ensures that all essential elements are included, properly formatted, and fully aligned with industry best practices and regulatory standards.
Automated Document Generation
The process of generating a protocol involves several steps that can be automated using AI:
Initial Draft Creation: Users provide a protocol synopsis, which the AI tool uses to generate the initial draft of the protocol. The synopsis includes key details such as study objectives, endpoints, and methodologies.
Text Generation: The AI tool automatically generates text for each section of the protocol based on the input synopsis. The AI can produce coherent and contextually appropriate text, ensuring that all necessary information is included.
Formatting and Structuring: The tool formats the generated content according to standardized guidelines, ensuring that the protocol is well-structured and compliant with regulatory requirements.
Collaboration and Version Control
After the initial protocol is generated, users can collaborate within the tool to edit and refine each section. The tool allows multiple users to access and edit the protocol simultaneously. Users can make iterative changes to the protocol, incorporating feedback and adjustments. The AI tool supports this process by tracking changes and maintaining version control.
Customization
Despite the use of standardized templates, AI tools offer flexibility for customization. Researchers can tailor the protocol to fit the unique requirements of their trial, combining the benefits of standardization with the need for specific adaptations.
The Impact of AI on Protocol Generation
AI-driven eProtocol generation represents a significant technological advancement in clinical trial protocol development.
By leveraging machine learning, utilizing standardized templates, and automation, these tools are advancing the creation of high-quality protocols, enhancing efficiency, and reducing errors.
Clinion’s AI: Transforming Protocol Design
Clinion leads the way in clinical trials, driving innovation with AIML and GenAI solutions. By utilizing GenAI tools, it can generate up to 90% of a full study protocol from just a brief synopsis, leveraging extensive training on real-world protocols. Our tool employs the standardized TransCelerate template to automatically generate and format each protocol section while enabling effective collaboration for reviewing and refining documents.
With these advancements, Clinion is not just streamlining trial processes; it’s setting a new standard for efficiency and accuracy in clinical research, ultimately enhancing patient outcomes and accelerating the journey to clinical breakthroughs.
External References

Abriti Rai writes on the intersection of AI, automation, and clinical research. At Clinion, she develops content that simplifies complex innovations and highlights how technology is shaping the next generation of data-driven clinical trials.
FAQS
Frequently Asked Questions
A clinical trial protocol is the official document that defines how a clinical study will be conducted. It includes objectives, eligibility criteria, study methodology, data collection procedures, and safety guidelines. It ensures the study is scientifically sound and regulatory compliant.
A poorly designed protocol can lead to trial delays, increased costs, low enrollment, and regulatory issues. A good protocol ensures clarity, consistency, and operational feasibility, improving trial outcomes and reducing the risk of failure.
An eProtocol is a digital version of the clinical trial protocol. It allows real-time collaboration, version control, and faster editing. When combined with GenAI, eProtocols can be created more efficiently and with fewer manual errors.
Gen AI automates protocol drafting by turning a brief synopsis into a structured protocol. It extracts key information and uses predefined templates, client examples, knowledge bases, and trusted sources to create accurate content.
Yes. While AI starts with standardized templates, users can customize any section to reflect the trial’s specific therapeutic area, study objectives, or regulatory requirements, ensuring flexibility without compromising structure.
Clinion’s Protocol Automation tool reduces drafting time by automatically generating up to 90% of the document. It enables teams to draft, collaborate, review, and finalize protocols seamlessly within a single unified platform.
Clinion ensures accuracy by grounding content in real-world protocols, a trained knowledge base, and trusted third-party sources. This minimizes hallucinations and maintains clinical relevance throughout the document.
With traditional methods, protocol drafting can take 160–220 hours. Using GenAI, a first draft can be generated in a day, with complete review and finalization taking just 1–2 weeks.
Clinion’s platform handles every stage, from AI-driven draft generation to collaborative editing, version control, stakeholder review, and final output. This streamlines workflows and eliminates the need for multiple disconnected tools.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Unlock the Future of Clinical Trials with Clinion.
Cut your trial costs by 35% and accelerate your time-to-market by 30%
Compliance
Fully Compliant with Global Standards


